Clinical Pipeline
Developing the next era of rapid-onset inhaled treatments
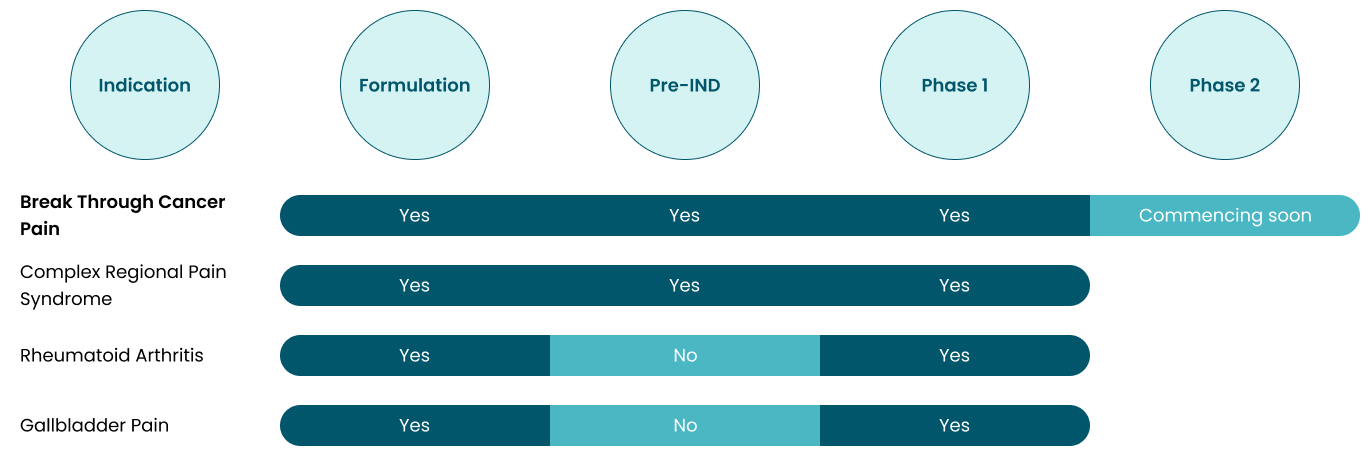
IRX-211
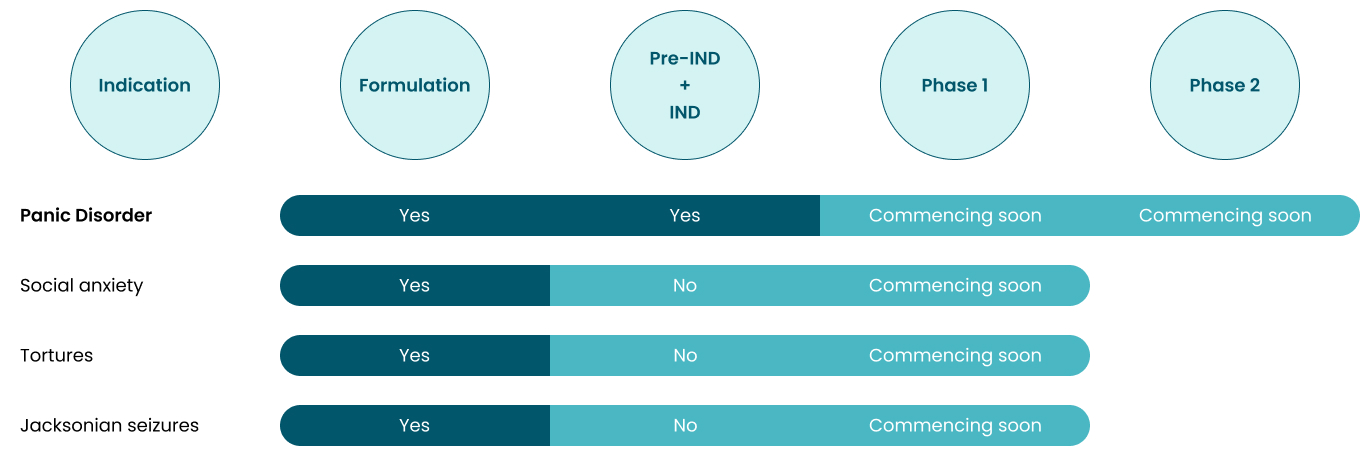
IRX-616a
Strategic Objectives
Short Term
Market Testing
- Acquire distribution experience in the Australian setting.
- Acquire valuable insights over 18 months of data from over 10,000 Australian patients and physicians to better understand the product market fit and the role of novel inhalation devices.
- Acquire Schedule 4, 8 wholesale / supply scheduled medications from the Department of Health.
- Acquire Import and Export licences from the Office of Drug Control (ODC).
Medium Term
New Product Development
- Work with best in class global experts for formulation configuration drug / device development plans.
- Source FDA compliant GMP and ISO13485 components for assembly.
- Test plume geometry and delivery efficiency.
- Commence a stability program.
Long Term
Commercialisation Following Clinical Trials
- Execute two drug development pipelines in parallel, both targeting an NDA via the streamlined regulatory pathway, FDA 505(b)(2) to develop the first ever FDA medically registered inhaled cannabinoid delivery systems.
- Acquire Approved Investigational New Drug (IND’s) on each drug candidate.
Regulatory Strategy
Faster approval process
Enabling access to use data from previously conducted studies of approved drugs can result in a faster approval process as the FDA.
Cost savings
There are often significant cost-saving opportunities as companies can leverage previously conducted studies of approved drugs, which can reduce the costs associated without executing a full drug-development program.
Reduced regulatory burden
The FDA may not require a full complement of preclinical and clinical studies.
Increased flexibility
The 505(b)(2) pathway provides opportunities to incrementally innovate on already approved drugs.

