IRX-211
Magnitude of Opportunity
Mental Health
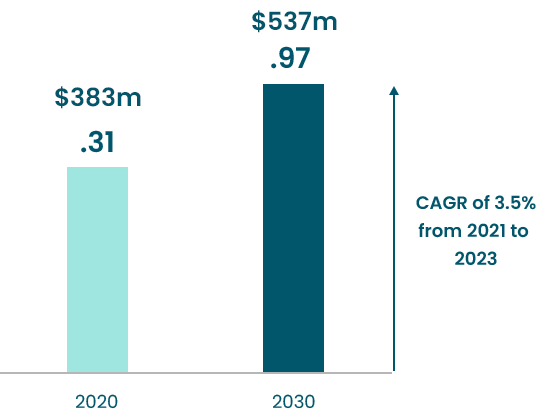
The global anxiety disorder treatment market is projected to reach USD 9 billion by 2030. during the forecast period 2022-2030. It includes panic disorders, post-traumatic stress disorder (PTSD), phobias, and obsessive-compulsive disorder.
- Panic Disorder is estimated to affect approximately 3-5% of the general population.
- More prevalent in women and typically begins in young adulthood. The exact prevalence of panic disorder is difficult to determine, as it is often under-diagnosed and under-treated.
GLOBAL ANXIETY DISORDER TREATMENT MARKET
$9
Billion (USD)
Assets under development – IRX-616a
Innovative Panic Disorder Therapy
IRX-616a is positioned to be the first FDA-approved inhaled medication for Panic Disorder, offering a novel treatment avenue.
Addressing an Urgent Need
With the COVID-19 pandemic increasing the prevalence of anxiety disorders by 25%, IRX-616a addresses a growing health crisis.
Unmet Market Demand
Existing treatments like SSRIs are plagued by side effects, such as disrupted sleep and increased suicidality risks, highlighting the need for alternatives like IRX-616a.
Unique Market Advantage
The lack of competition in inhaled medications for Panic Disorder strengthens IRX-616a’s commercial prospects.
Large Market Opportunity
The total addressable market for anxiety and depression treatments is projected to reach $13.3 billion by 2027.
Strategic Regulatory Plan
An IND has been submitted, and FDA approval would lead to further approvals by the EMA and TGA, enhancing its global reach.

